Shedding light
By Lori Ann White
Light sources are the ultimate killer apps for particle physics technology. Their brilliant X-rays illuminate every aspect of the material world, from the inner workings of cells to the intricate dance of the electrons that create chemical bonds. As these all-purpose scientific tools evolve, the payoffs include better batteries, greener energy, new high-performance materials, more effective drug treatments, and a deeper understanding of nature.
 |
| Researcher Wanli Yang probes the electronic structures of battery materials with soft X-ray fluorescence at Berkeley Labs Advanced Light Source. The LCD screen in front shows the experimental setup inside the ultra-high vacuum chamber; the nose cone of the detector points to samples mounted on a vertical manipulator and exposed to the soft X-ray beam. Photo: R. Kaltschmidt, Lawrence Berkeley National Laboratory |
In 1971, physicist Burton Richter of Stanford Linear Accelerator Center was building a new type of particle collider called a storage ring. The labs two-mile-long linear acceleratorhoused in what was then the longest building in the worldwould shoot electrons and their antimatter twins, called positrons, into the 80-meter-diameter Stanford Positron Electron Accelerating Ring, and SPEAR would set the beams of particles on a collision course. Richter and his colleagues stood by to examine the debris to see what discoveries came out.
Physicists were already familiar with one product of accelerating particles in a circle at close to the speed of light: synchrotron radiation. To high-energy physicists, synchrotron energy was an annoyance, an unwanted demonstration of the laws of physics in this case, a law that says charged particles radiate energy when accelerated. Synchrotron radiation would rob electrons and positrons in the SPEAR ring of energy that would better serve science by going into the creation of new particles and the illumination of new physics.
Two Stanford faculty members, condensed-matter physicist Sebastian Doniach and electrical engineer William Spicer, had a different vision. They saw in the SPEAR storage ring a new and better way to satisfy an already huge demand for X-rays. They saw a source of high-energy electromagnetic waves whose short wavelengths and highly penetrating nature could illuminate materials on a scale small enough to show atomic structures and reveal electronic properties.
Doniach and Spicer approached Richter even before SPEAR was finished.
They said that they would revolutionize condensed-matter physics if only I would let the X-ray beam out of the storage ring. That was their pitch, Richter recalls.
After a quick, informal inquiry to see if the two professors were blowing smoke, as he puts it and obtaining assurances that modifying the ring would not endanger its high-energy physics mission Richter said, Lets do it.
The SPEAR construction team removed a section of the ring and modified it, adding a beam port to let the X-rays out. And Richter did Doniach and Spicer another favor: I bought them a Sears garden shed to use for experiments.
The Stanford Synchrotron Radiation Project was under way.
 |
| Some early experiments at the Stanford Synchrotron Radiation Project took place in a Sears garden shed mounted on the concrete roof of the SPEAR particle storage ring. Ian Munro (third from right, wearing a tie), who was visiting that year from the NINA synchrotron in England, was the key scientist in the development of this experimental station. He provided haggis and drinks to celebrate first light. On Munros right are Ben Salzburg, Axel Golde, Sebastian Doniach (first director of SSRP) and George Brown. Photo: SLAC Photo Archives |
As promised, the stronger, more intense X-rays streaming into the garden shed began making their mark on condensed-matter research and structural biology. Stunningly clear data soon proved the value of synchrotron radiation for investigating the chemical compositions and electronic structures of materials, while the first images of protein structures using such strong X-rays soon followed. Both breakthroughs came as Richter continued to smash together electrons and positrons; the X-ray experiments may have been parasitic on the main program, but the parasites did not disturb their host.
In 1974, Richter and his team discovered the J/psi particle, for which he shared the 1976 Nobel Prize in Physics.
Today, more than 70 light sources are in operation or under construction around the world, including the descendent of that original project, the Stanford Synchrotron Radiation Lightsource or SSRL.
No longer an afterthought, dedicated light sources are critical research tools for everything from developing better batteries to finding the causes of disease. They contribute to the development of new medicines. They delve into the structures of proteins, the secrets of photosynthesis, the behavior of catalysts. Developing high-performance materials, investigating pollutionany time a researcher wants to spy on a process or unravel a structure, if its on a molecularor now atomicscale, a light source is the tool of choice.
As Richter points out, Doniach and Spicer were simply too modest when they said they were going to revolutionize condensed-matter physics. They revolutionized more than that. Having a million times more intense X-rays gave you capabilities that nobody ever dreamed of being able to exploit.
Illuminating proteins
 |
| Lisa Keefe at one of the two Advanced Photon Source beamlines she oversees at Argonne National Lab. The Industrial Macromolecular Crystallography Association, a consortium of major pharmaceutical companies, designed, built and staffed these beamlines. IMCA member companies do research there on potential drug treatments. Photo courtesy of L. Keefe, Industrial Macromolecular Crystallography Association |
Those million-times-more-intense X-rays have transformed the study of structural biology, enabling scientists to get the first good look at the intricate cellular machines that build the structures of our bodies and generate the energy that powers us. True, researchers had deciphered some protein structures as far back as the late 1950slong before the widespread use of light sources. However, by 1973, two years after being established and just as SPEAR was coming on line, the Protein Data Bank held just nine structures. Today the Protein Data Bank, a key resource for structural biologists the world over, holds the structures of more than 72,000 proteins.
The revolution did not happen overnight. At first, the primitive state of imaging technology made high-resolution results very hard to get. The protein crystals themselves were the biggest stumbling block.
As Bob Sweet, a beamline scientist at Brookhaven National Laboratorys National Synchrotron Light Source, puts it: You have to understand that protein crystals are the texture of firm Jell-O and very moist. They dont do very well in an X-ray beam.
Two breakthroughsfreezing the crystals to retard radiation damage, and the development of better detectors using CCD chipstransformed the use of synchrotron light sources, according to Thomas Steitz of Yale University, who shared the 2009 Nobel Prize in Chemistry for unraveling the structure of the ribosome, the cellular factory that builds proteins from genetic instructions brought by messenger RNA.
It would have been impossible to have solved the ribosome without a synchrotronperiod, Steitz says. In my opinion its a good thing there are light sources scattered everywhere.
Discovering drugs
Academic crystallographers such as Steitz generally focus on determining the structures of biologically important proteins. Once those structures are unraveled, crystallographers from pharmaceutical companies home in on receptor sites on the proteins where disease-causing agents can latch on and wreak havoc. Thats the best place to ambush and disable them.
The techniques for studying these receptors are simpler to execute and faster to complete than determining the structure of an entire protein from scratch. But finding the right drug can still take years. Continually competing for access to beamlines at national facilities just wont work for pharmaceutical companiesnot if they want to get anything accomplished yet light sources are generally national facilities for a reason. Theyre big, complicated, and full of sophisticated machinery requiring highly specialized knowledge to maintain and operatein other words, expensive. Very expensive.
Thus was born the Industrial Macromolecular Crystallography Association, or IMCA, a consortium of pharmaceutical companies that approached Argonne National Laboratory during the construction of the Advanced Photon Source. IMCA members would provide the money to build, equip, staff, and run beamlines; Argonne would provide the X-rays. The result is Sector 17 at the APS, two specialized beamlines dedicated to the fast turnaround of candidate substances for drugs.
Its rather remarkable that all the companies manage to cooperate, says Lisa Keefe, director of the IMCA Collaborative Access Team program at the APS. They are all competitors. One reason the collaboration works: their work remains proprietary. Membership varies, but currently consists of Abbott, Bristol-Myers Squibb, Merck, Novartis and Pfizer, with subscription programs available to companies that are not IMCA members. As part of the deal with Argonne and the Department of Energy, the beamlines are also open to general users 25 percent of the time, with general referring to anyone who comes up with a good proposal. In practical terms, this means academic research groups.
Time is money to these companies, and their money has bought them time. According to Keefe, the beamlines are focused on very-high-throughput screening and data collection. With the state-of-the-art instrumentation at beamline 17-ID, researchers can collect a data set every three to five minutes.
One of the first members of IMCA, pharmaceutical giant Abbott Laboratories, used the APS to study HIV, the virus that causes AIDS. That research directly contributed to the development of Kaletra, a highly effective protease inhibitor that stops the HIV virus from replicating. GlaxoSmithKline relied on research conducted at the APS to create Votrient, a treatment for kidney cancer, and Merck developed Januvia, a treatment for diabetes.
Bristling with beamlinesAt SLACs SSRL and other synchrotron light sources, electrons speeding around a ring emit intense light, usually in the form of X-rays. The light streams out of the ring into beamlines equipped with specialized instruments for conducting a wide variety of experiments. This diagram shows the types of research taking place at each SSRL beamline. The dotted lines show planned future beamlines. Techniques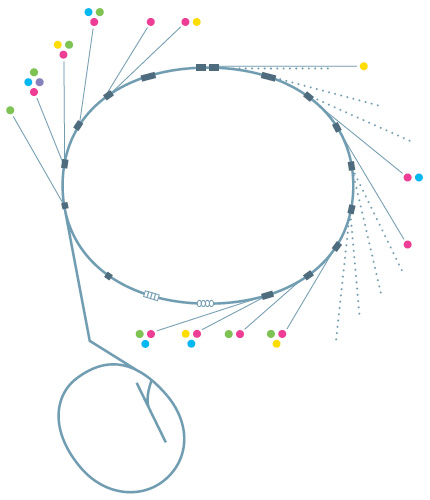
|
Free electron laser
Electrons slalom through the paired magnets of an undulator in a free-electron laser. The back-and-forth motion causes them to emit synchrotron radiation. Unlike the synchrotron light emitted by particles going around a storage ring, the radiation from FELs is coherent, with the crests and the troughs of the light waves lined up; coherence gives it much more intensity. Image: Sandbox Studio |
Research at the SSRL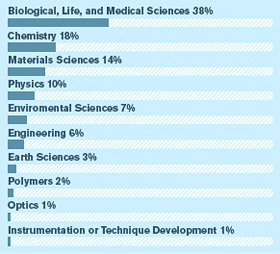 |
Basic research at light sources has also given rise to start-up companies aimed at turning discoveries into effective treatments. Steitz co-founded Rib-X Pharmaceuticals, a company that develops more effective antibiotics based on the structural information about the ribosome he gathered at light sources. A Rib-X antibiotic that has made it through phase II clinical trials is designed to combat methicillin-resistant Staphylococcus aureus, or MRSA, an antibiotic-resistant bacterium that kills thousands annually.
In California, Stanfords Roger Kornberg, who won a Nobel Prize for determining the structure of a key enzyme involved in translating DNAs genetic instructions into proteins, founded a company called Cocrystal Discovery to find drugs that block this process in RNA viruses like the ones that cause influenza and hepatitis.
Cocrystal is carrying out studies at a number of DOE light sources, including Argonnes APS and the two facilities where Kornberg performed his prize-winning researchSLACs SSRL and the Advanced Light Source at Lawrence Berkeley National Laboratory.
I believe the whole future of drug development lies in synchrotrons, says Kornberg, who heads a research lab at Stanfords School of Medicine.
Light sources vs. disease
 |
| Top: A series of images from research at the National Synchrotron Light Source. 1) Amyloid plaques associated with Alzheimers disease in human brain tissue, fluorescing in green; 2) zinc and 3) copper ions in the same tissue sample; and 4) an overlay of the previous three images reveals that the plaques contain high levels of the two metals. Bottom, from left: Lisa Miller, Andreana Leskovjan, and Tony Lanzirotti at one of the NSLS beamlines at Brookhaven National Lab where they conducted their Alzheimers research. Images and photo courtesy of L. Miller, Brookhaven National Laboratory |
Lisa Miller uses Brookhavens NSLS, the first facility built expressly for research using synchrotron radiation, to study diseases themselves. Two favorite targets are Alzheimers disease and amyotrophic lateral sclerosis, also known as Lou Gehrigs disease.
Miller and her research group have made good progress in understanding the cause of Alzheimers using synchrotron light sources, although at this point in her research, progress means the ability to ask more pointed questions.
They use two different types of synchrotron-based microscopes to examine the Alzheimers-afflicted brain, Miller explains. Synchrotron infrared light reveals organic materials like proteins and lipids. With synchrotron X-rays, Miller can identify traces of metals like copper, iron, and zinc.
Miller uses infrared microscopy to study the structure of amyloid plaques, the tell-tale clumps of misfolded protein that mark the brains of Alzheimers victims. For some reason, these plaques are toxic and kill brain cells, she explains. The brain of an Alzheimers sufferer can shrink to 60 to 70 percent of its original size.
Using X-ray microscopy, Miller has discovered that human plaques are loaded with metal ions, especially copper and zinc. But interestingly, mice that are genetically engineered to develop Alzheimers plaques do not have metal in their plaquesand their brain cells do not die.
Is it the metal that causes plaques to be toxic? And how does the metal get there in the first place? she asks. You dont have free copper just floating around in your body. That would be toxic.
To answer her questions, Miller wants to get a closer look at the sticky plaques and trapped ions. Shes eagerly awaiting the National Synchrotron Light Source II, which will generate X-rays more than 10,000 times brighter than those of its predecessor. It will have a resolution of tens of nanometers, she says. Well be able to see individual neuronsor even individual mitochondria within the neurons. She wants to examine whats there even before the plaques form.
In the meantime, shell continue to put the NSLS to good use for as long as she can.
My fear is that the bad budget situation will cause the NSLS to turn off before NSLS-II turns on, she says. If that happens, Lisa Miller will truly find herself in the dark about Alzheimers disease.
Building better batteries
At Berkeley Labs ALS, scientist Wanli Yang and his colleagues are developing better electrode materials for lithium-ion rechargeable batteries. Currently these batteries are the most popularand most promisingrechargeable battery technology, but theres definitely room for improvement.
For example, graphite, a form of carbon commonly used in lithium-ion battery anodes, is actually not the most efficient material for energy storage. Other substances, such as silicon, have a much higher energy capacitythey can hold more lithium ions when a battery is charged.
But researchers have already learned that replacing the graphite with silicon leads to other problems. When lithium ions flow into a silicon anode during charging, the volume of the anode expands almost three-fold. When the battery is discharged, the ions flow out of the anode and the silicon is left adrift, leading to broken mechanical and electrical connections that ruin the battery.
Collaborating with material chemists and theoreticians, Yangs group has been using the X-rays of the ALS beamline to study versatile polymer materials that could glue the molecules of the silicon anode together while maintaining good electrical conductivity, thereby enabling the best of both worldshigh energy capacity and the ability to withstand numerous recharges.
A more fundamental goal is to reveal crucial electronic structures that actually define battery performance, thus providing a basis for the development of even higher-performance materials for use in future batteries.
Yang reports progress on this front, as well. The collaboration has pinned down a particular electronic state of the polymer, what Yang calls a very special energy for electrons to stay with, thats key to maintaining efficient battery operations. This electronic state can be measured directly by soft X-ray spectroscopy, and, says Yang, such synchrotron-based direct probes turn out to be a very powerful method for studying other battery materials, as well.
Synchrotron-based X-ray spectroscopy is the only technique that can tell us all the detailed information we need, says Yang. Its chemically sensitive, sensitive to the orbitals of the electrons. By using soft X-ray spectroscopy, we can see where the electrons are and where they go.
Can we do what plants do?
 |
| Berkeley Lab staff scientist Jinghua Guo uses the labs Advanced Light Source to study the electronic structure of a catalyst designed to promote a chemical reaction involved in artificial photosynthesis. In situ studies like these, which take place in realistic operating environments, play an important part in designing new materials with improved qualities. Photo: R. Kaltschmidt, Lawrence Berkeley National Laboratory |
If only we were like plants. Plants dont have an energy crisis and wont for another five billion years. Thats becauseas we learned in schoolplants pull all the energy they need from the sun, converting light energy to chemical energy by turning carbon dioxide and water into sugars and releasing oxygen as a waste product.
Plants have it made.
But what if we could do the same, in a sense convert sunlight to chemical energy of a form more suited to our industrialized society, such as biofuel?
Wed have it made.
Thats the motivation for the Joint Center for Artificial Photosynthesis (JCAP), a collaborative effort among several California universities and national laboratories thats funded by the US Department of Energy.
Jinghua Guo is a scientist at the ALS, at Lawrence Berkeley National Lab, whichalong with SLAC and the University of California at Berkeleyconstitutes the northern California JCAP site. According to Guo, The idea behind JCAP is to discover a system that mimics naturebut first you have to understand what happens in nature.
Guo and other JCAP scientists use soft X-rays from the ALS to watch natures chemistry happen. Generally that means watching electrons move around, or even arranging them himself and then watching what happens. You can tune soft X-rays so precisely that you can put an electron where you want, he said.
One system Guo is investigating with other JCAP scientists consists of a cluster of titanium, manganese, and cobalt atoms. Put the cluster in solution, says Guo, and it will effectively absorb sunlight and release oxygen from water. Theyre trying to unravel the details of that reaction.
Experiments like these, which look at how materials function in realistic operating environments, are a tricky but important part of an approach known as materials by design, said Zhi-Xun Shen, SLACs chief scientist and head of the Stanford Institute for Materials & Energy Science.
It starts with using theory and computer simulations to design a new materialwhether a catalyst, a battery component, or a novel superconductor that carries electricity with 100 percent efficiency. Scientists go into the lab and make samples of the material, then use X-rays from a light source to determine its structure and chemical composition and watch it perform under realistic conditions. Catalysts go through more rounds of testing to see how active and selective they are in promoting specific chemical reactions.
SLACs Anders Nilsson, who is deputy director of SUNCAT, the Center for Sustainable Energy through Catalysis, has a particular interest in surfaces; thats where catalysts do their work of helping chemical reactions happen. Recently he led a research team that used the labs SSRL to get a good look at a platinum catalyst in the act. They created a sample of platinum one layer thick, hit it with a wavelength of X-ray light that was tuned to interact with the platinum and only the platinum, and captured what happened with a recently developed and highly sensitive type of X-ray spectroscopy called high-energy resolution fluorescence detection.
Capturing what happens to a sample in a realistic environment enables researchers to connect theory to reality, Nilsson says, adding, X-rays have a really great potential for illuminating surface chemistry, which can give us insights into the development of new materials.
The future: bright and brighter
 |
| Top: An X-ray laser pulse from the Linac Coherent Light Source hits a molecule and scatters light into a detector, forming a diffraction pattern. By combining many diffraction patterns (bottom left), scientists recreate the proteins 3D structure (bottom right) Images: G. Stewart, SLAC National Accelerator Laboratory |
The Stanford Linear Accelerator Center is now SLAC National Accelerator Laboratory. SPEAR3 is still going strong, running 24 hours a day nine months of the year, serving 1400 users annually at more than two dozen experimental stations.
As for the famous 2-mile-long linac? Fittingly in the era of reuse, recycle, and reduce, the lab has repurposed it to accelerate electrons for the Linac Coherent Light Source. The LCLS is the premier example of the newest generation of light source, the free-electron laser. FELs, with their pulses of X-ray laser light, can see even smaller objects and follow even faster reactions than can regular synchrotron radiation sources.
The LCLS, the first of the hard X-ray FELs, is a billion times brighter than the brightest conventional light source. It packs all its energy into laser pulses of less than 1/10th of one trillionth of a second. These ultrafast pulses promise to take portraits of atoms, not just molecules, and to capture chemistry in the act.
Keith Hodgson is SLACs associate laboratory director for photon science and the labs chief research officer. Hes been a researcher at the lab since the pioneering days of SSRP. Hodgson points out that the LCLS owes a special debt to high-energy physics. Getting the electrons to lase required exquisite control of the electron beama 5-micrometer beam over long distances, with high precision, he notes. Only accelerator physicists trained to meet the exacting needs of particle physics experiments could have done it.
Two papers appearing recently in the journal Nature hint at the LCLSs capabilities. In one experiment, a team of researchers captured single-shot images of an entire uncrystallized Mimivirus, the largest known virus. In the second experiment, the same team confirmed the molecular structure of a hard-to-crystallize protein called Photosystem I, an important protein complex involved in photosynthesis, using nano-sized versions of the crystal that are easier to prepare. If it lives up to its initial promise, the technique could cut years off the analysis of some proteins.
Still ahead: molecular movies of chemical processes that happen too quickly to capture with conventional X-rays.
Forty years ago Sebastian Doniach and William Spicer told Burton Richter they would revolutionize condensed-matter physics if Richter would let the X-rays shine.
The X-rays are shining brighter than ever, and the revolution continues.
Click here to download the pdf version of this article.