|
|
| Illustrations: Sandbox Studio |
As a particle physicist, Alan Litke routinely measures tiny signals with equally tiny electronics. Now hes applying those methods to individual nerve cells, revolutionizing the study of how we see.
By Lizzie Buchen
Seeing is easy. We open our eyes, and there the world is–in starlight or sunlight, still or in motion, as far as the Pleiades or as close as the tips of our noses. The experience of vision is so common and effortless that we rarely pause to consider what an astounding feat it is: Every time our eyes open, they encode our surroundings as a pattern of electrical signals, which the brain translates into our moving, colorful, three-dimensional perception of the world.
This everyday miracle has attracted the devotion and expertise of an unlikely individual–Alan Litke, an experimental particle physicist based at the University of California, Santa Cruz. When not in Geneva, Switzerland, where he is working on the ATLAS particle detector for the Large Hadron Collider, Litke is working with neuroscientists and engineers, adapting the technology of high-energy physics to study the visual system.
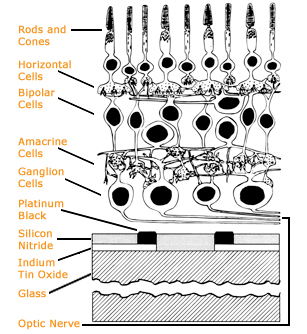
The central challenge is to understand the language the eye uses to send information to the brain. Light reflected from our surroundings enters our eyes through the transparent window of the cornea and is focused by the lens, forming an image on the retina. The retina of each eye contains about 125 million light-sensitive rods and cones, which translate light into electrical and chemical signals. These signals travel to the visual centers of the brain through a million retinal ganglion cells, or RGCs.
The retina thus encodes the activity of 125 million cells in the signals of one million output cells, which deliver the brain a highly compressed neural code from which our entire visual experience is derived. Litke wants to understand how this neural network processes information from our surroundings and portrays it to the brain.
Coming from a particle physics background presented many challenges for Litke. Not only would he need to adapt particle detector technology for the messier, wet world of living tissue, but he would also need to win over skeptical biologists and funding agencies. He was proposing a whole new way of doing research in neuroscience, one that promised a vast leap forward in what could be measured and analyzed.
Litke's interest in neuroscience began with his daughter's wobbly first steps. At the time, he was developing the first silicon microstrip detector systems for the Stanford Linear Accelerator Center's MARK II experiment. These systems consist of many very narrow detecting strips, fabricated on a thin silicon wafer, which record the passage of subatomic particles; when read out with specially-designed integrated circuits, they can deliver their vast amount of data over just one line, instead of a nest of wiring. The goal of the project was to detect the charged particles produced in Z boson decays with unprecedented spatial resolution, but the real object of his fascination was the technology itself. “It was marvelous,” he recalls. “I really loved that technology.”
As he watched his daughter teeter along, he marveled at how her developing brain adapted to the novel, bipedal world. “I had started reading a little about artificial intelligence, and I thought, ‘ This can't be how the brain works!’ I couldn't imagine my beautiful daughter learning to walk if her brain was a set of if/then statements, purely logical. It's much more magnificent and beautiful than that.” He adds, “I didn't know much about the brain, but I knew that if you wanted to understand it, you need to get in there and really see the circuitry. I kept thinking about this incredible technology we were working with, and I wanted to come up with a way to use it for the brain.”
Litke appealed to his group at SLAC, trying to lure them into his neurobiology vision, but there were no immediate takers.
Meanwhile, Markus Meister, a postdoc in Dennis Baylor's neurobiology lab at Stanford University, was leading groundbreaking experiments on the retina.
An appealing slice of tissue
The retina appeals to scientists studying neural circuitry for a number of reasons: All the input neurons–the rods and cones–are known, as are a number of its output neurons, the retinal ganglion cells. The input signals can be easily controlled just by shining light on the retina. And the output signals can be easily monitored, in principle, by recording the electrical activity of the RGCs with electrodes. Further, what scientists learn from studying the retina can be applied to understanding the function of any neural circuit–a central goal of neuroscience.
For decades, studies of neural function in the retina and brain were restricted to recordings from single neurons. It was presumed that these measurements could be pieced together to decipher the functions of complex circuits, but Meister wasn't convinced; he believed it would be necessary to record from many neurons simultaneously.
Meister had already started working with a 61-electrode array, originally developed by Jerry Pine, formerly a particle physicist at SLAC. But he needed more help. As luck would have it, Meister's neighbor was a postdoc in Litke's lab and arranged an introduction.
“It seemed to me like a wonderful project,” Litke recalls. “To a physicist, the retina is like a particle detector. It's an advanced pixel detector that detects light, and converts it to an electrical signal. I knew the only way to figure it out was to record from live retinal tissue.” As Meister developed the methods for monitoring the simultaneous electrical activity of many neurons, Litke volunteered to contribute in any way he could. He started to help with the electrode array fabrication, and published a paper with Meister in 1991.
|
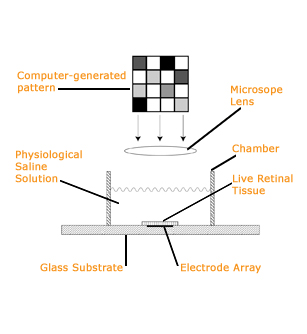
The technique involves placing a slice of retinal tissue on top of the array in a chamber filled with a special solution that can keep the tissue alive for several hours. Images are then focused on the retina's photoreceptors while the electrodes monitor the responses of the retinal neurons. At the time, an array with 61 electrodes was revolutionary and, today, is still considered state of the art. But Litke had higher aspirations.
“In physics, when you design a new instrument, like a new accelerator, you want to go up by a factor of 10 in energy, in resolution, whatever it is,” he says. “So, not really knowing what the scale was for interesting neurobiology, I thought, ‘ We get tens of neurons now; let's go up to the hundreds.’ A factor of 10 seemed like an interesting step, and it seemed more appropriate for the level of information the retina was feeding to the brain.”
But Litke's vision wasn't embraced by his collaborators. “They were still learning to graduate from one to 10, so more would be a big leap, and I couldn't convince them it was worth doing,” he says. “Without the support of the biologists, we couldn't get funding.”
Litke then moved to Geneva to devote himself to high-energy physics, visiting California only occasionally to lobby for the next-generation retinal measurement device. He had all but given up when he received a call from Bob Eisenstein, head of the physics division of the National Science Foundation. “I assumed he called to talk about physics,” Litke says, “but it turned out he wanted to talk about neurobiology.”
Cultures collide
Eisenstein had been trying to push biological physics within the NSF and had heard about Litke's work with Meister. As Litke recalls, “He had a call for proposals but didn't receive anything interesting, so he wanted to hear more about my work. I took the proposal very seriously, and faster than any proposal I've ever submitted, it was approved.”
Finally, Litke had the financial resources and encouragement to pursue neuroscience once again. He returned to his original goal of developing arrays of electrodes that would record from hundreds of neurons simultaneously. “To biologists, using this many electrodes to record from live animals was inconceivable–they didn't see how it was technically possible,” Litke says. “But to me, we were doing this daily at CERN!”
Litke assembled a team from the high-energy physics community. His first ally was Wladyslaw Dabrowski, a physicist and integrated circuit designer from the AGH University of Science and Technology in Krakow who had been working on read-out chips for ATLAS. To begin, the team made prototype 61-electrode array systems that were smaller, denser, and more advanced than the ones Meister had been working with. The goal was to eventually develop an array with 512 electrodes.
But when Litke asked about more funding from the National Institutes of Health, he was strongly discouraged. “Basically, the program manager said I wasn't really doing anything, just building equipment,” Litke says. “They wanted a hypothesis. They didn't want instrumentation.”
Litke was shocked. In the world of physics, technology development is recognized as vital for new discoveries. But the life sciences are more hesitant about exploring something completely unknown, and thus a well-founded hypothesis is required. “I couldn't believe it. This technology would take neurophysiology to another realm!” Litke says. “It would answer questions that cannot be addressed by current technology. It's an incredible story to me as a physicist.”
At the time, Litke was working full-time on the ALEPH experiment at CERN, while spending nights and weekends working on his neuroscience arrays. He continued making trips to Stanford to talk with Baylor and his postdocs, who were working with Meister's 61-electrode array.
Although most of the postdocs were unwilling to advance past 61 electrodes–the technology's possibilities had certainly not been exhausted–one, E.J. Chichilnisky, was captivated. Eventually, Baylor also became convinced, and wrote an influential letter of support to the NSF, generating further funds for Litke's project.
“Most people weren't interested because they didn't see the point,” Chichilnisky says. “We didn't have enough information from our 61-electrode arrays to know whether it was worthwhile to go to another level. It was risky.” Yet Chichilnisky was excited about the project, and confident of its significance: “The truth of the matter is I don't know why. It was a gut feeling.”
A groundbreaking leap
When Chichilnisky took a faculty position at the Salk Institute in La Jolla, California, in 1998, he began collaborating with Litke, using the prototype 61-electrode version of a new, more advanced array to help evaluate the function of live retinal tissue.
“These chips were completely different than the original 61-electrode arrays that Meister was using,” Litke says. “We completely redesigned everything. We needed it to be high-density, with many interconnected channels. Everything was inspired by silicon microstrips.” The geometry was different, but the concepts were all direct from the Mark II Silicon Strip Vertex Detector.
The first 512-electrode array went into use in 2003.
Litke says, “When biologists saw this, they were flabbergasted. When they think of 512 electrodes, they think of 512 cables coming out, a big amplifier, a room filled with electronics. When they saw this tiny array–hundreds of electrodes, all squeezed into 1.7 square millimeters on a small printed circuit board, and one little cable–they were really excited.”
Chichilnisky says the unique technology has revolutionized his work, allowing his lab to examine, with unprecedented power and resolution, how patterns of RGC activity interpret the visual world for the brain. While focusing on specific aspects of visual perception, such as motion and color, he is also developing models that would allow one to predict and reproduce RGC activity from the visual stimulus alone, an accomplishment that could contribute to the development of prosthetic devices for the visually impaired.
For Chichilnisky, the ability to monitor the activity of hundreds of RGCs simultaneously was initially the biggest draw. But in 2007 a new reason emerged, leading to the group's biggest discovery yet.
Among the one million RGCs “there are something like 20 different types of ganglion cells,” Chichilnisky explains, “each of which is distinct and conveys different types of information. But less than half have really been studied, because they're so rare you can't detect them with traditional techniques.” The various types of cells form parallel visual pathways that communicate contours, movement in specific directions, and colors as separate images for the brain to piece together. To gain a comprehensive understanding of the information the brain receives, it is vital to understand what each of the 20 types does.
A 61-electrode array doesn't have enough coverage to do that. However, with a 512-electrode array, the researchers could distinguish each type of cell and its function, Chichilnisky says: “You get a completely new level of clarity about all the visual signals.”
This clarity led to a groundbreaking finding that established the value of Litke's device as a tool in neurobiology. In a paper published in October 2007 with Dumitru Petrusca–a physics student who had developed software for ATLAS–as the lead author, Litke, Chichilnisky, and their team reported the discovery of a new class of RGCs in the primate retina, thought to help primates detect motion. They named it the “upsilon” cell. “They've been searching for it in primates for over 40 years,” Litke says. “It's such a small fraction of all the ganglion cells, so it was impossible to confidently detect with single- or even 61-electrode techniques. But when we recorded with this array, we'd get five to 10 upsilon cells, so we knew it wasn't an artifact.”
 |
| In the center of this chip, an array of 512 electrodes no bigger than the head of a pin records signals from neurons that transmit information from the retina.
Photo courtesy of Alan Litke |
Pushing the limits
When Litke and Petrusca started to write the paper, they encountered another major difference between physics and neuroscience. “I was hoping I could just write about our methods and present the data,” Litke says. That's how it works in physics–there are so many new devices and techniques that researchers typically just reference the most recent and most relevant. But in neuroscience, the publishing culture requires references going back nearly a century at times. He also had to get used to sensitivities involving the order in which authors are listed on a paper. The average neuroscience paper typically has fewer than six authors, Litke says, and the order matters when people are trying to get jobs. In high-energy physics, on the other hand, collaborations of several hundred simply use alphabetical order.
Authorship of papers is not the only difference between particle physics and neuroscience culture. Litke's experience with the highly collaborative nature of particle physics has influenced his neuroscience labs. “He changed the atmosphere here,” says Jeff Gauthier, a graduate student in Chichilnisky's lab. “In most neuroscience labs, everyone is working on their own project and is very independent from one another. But the experiments with Alan's array will only really work if everyone in the lab helps each other out. We have our own projects, but in order to maximize the use of the technology and the animal tissue, we all work on each others' projects, too.”
Encouraged by progress in Chichilnisky's lab, Litke decided to expand his neuroscience work at the University of California, Santa Cruz, where he was still working full-time on ATLAS. But it continued to be a struggle: “We didn't have a lab, we didn't have animals to work with, and even getting a postdoc to work on the project was a challenge, because the work was so risky. You come from a field where you know a lot, and enter one in which you know virtually nothing.” Litke was eventually able to convince high-energy physicist Alexander Sher to join his neuroscience crusade as a postdoc. “We talked about whether it would be better to continue in high-energy physics and work with ATLAS,” Sher says. “But with neuroscience, I'd be part of a small team, doing groundbreaking work. I really got into the biology.”
The reach of Litke's technology now goes beyond the retina. He has ongoing or proposed projects to study the brain activity of naturally behaving barn owls and rats to try to understand the connections between their behaviors and their neural activities. Nevertheless, he is still frustrated by funding issues. “With neuroscience proposals, you have to start out by saying how your research is going to help autism or Alzheimer's disease and such,” Litke says. “I can't just talk about how wonderful the technology is, and all the potential it holds. Everything has to be low-risk. I learned from the biologists that you only propose to do things you've essentially already done.”
Litke doesn't think he'll be able to spread himself between physics and neuroscience much longer. “It's getting to the point where I'm going to have to decide on one field, and the truth is I don't know which it will be,” he says.
Still, he is reveling in the possibilities before him: Stick with the ATLAS collaboration to help open a new era of particle physics, or move full-time to neuroscience and try to answer the questions raised by watching his daughter start to comprehend the world. Either way, he'll be pushing the limits of detector technology to measure and probe, in search of the answers to the most fundamental questions of science.
Click here to download the pdf version of this article.